Retinal organoids to predict the effectiveness of gene therapies
Researchers at the Institut de la Vision evaluated the effectiveness of CRISPR-Cas9 gene editing on retinal organoids compared to models commonly used to develop gene therapies. Their publication*, published in December 2025 in the journal Molecular Therapy Methods & Clicival Development, shows that these lab-grown 3D structures faithfully replicate the results obtained in vivo, paving the way for safer and more effective gene therapies for inherited retinal diseases.
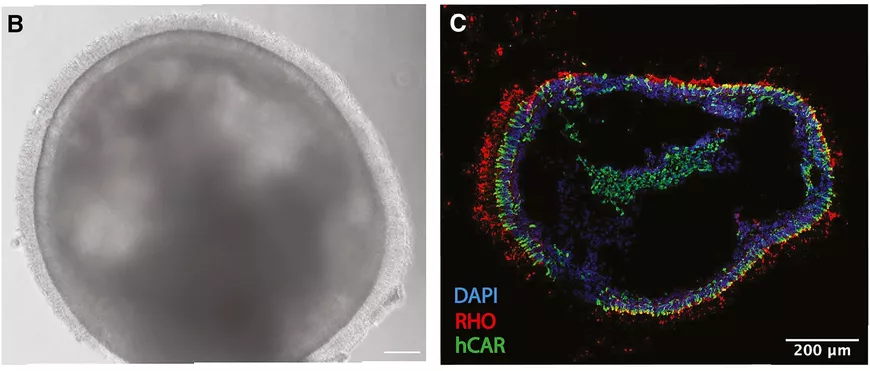
(B) A 150-day retinal organoid, the presence of cilia on its surface indicates the presence of photoreceptors. (C) Cryosection of a retinal organoid. In red, rhodopsin, an indicator of the presence of rods; in green, arrestin, an indicator of the presence of cones.
The challenge of gene therapies for the retina
Retinitis pigmentosa are inherited diseases caused by mutations in more than 90 different genes, leading to the dysfunction or death of photoreceptors. To restore vision in affected patients, the most promising avenue is that of gene therapy, which aims to correct or compensate for the defective gene. While some successes have been achieved, the diversity and complexity of retinitis pigmentosa make global approaches difficult. Therapies using genome editing tools, such as CRISPR-Cas9 molecular scissors, could offer personalized therapeutic solutions, tailored to each mutation. However, classical experimental models do not allow to faithfully reproduce the genomic and structural environment of diseased human photoreceptors. This lack of an appropriate model makes it uncertain whether potential therapies will go into the clinic: it is difficult to accurately predict their effectiveness in humans.
From the Cell to the Retina: Organoids Pass the Test
To accelerate research, the Gene Therapies and Models of Neurodegenerative Diseases team, led by Deniz Dalkara at the Institut de la Vision, is focusing on a fast-growing experimental model: retinal organoids. These tiny, three-dimensional biological structures, just a few millimeters in size, are much closer to what happens in our bodies than conventional two-dimensional cell cultures. Even if they do not exactly reproduce an organ, organoids offer much deeper access to biological processes and their pathological disorders. It is even possible to create organoids from patients' cells, for example by taking cells from their skin and reprogramming them into induced stem cells (iPS). This approach faithfully reproduce a patient's specific genomic and physiological environment – with its mutations – which is particularly valuable in the development of gene therapies against retinitis pigmentosa.
In order to explore the potential of retinal organoids in this context, Juliette Pulman and Hugo Malki, researchers in Deniz Dalkara's team, compared their response to CRISPR-Cas9 genome editing to classical models. For their study, they used 150-day-old retinal organoids from skin cells from a healthy donor. The advanced maturity of these organoids allows them to develop structures naturally present in the retina, including functional photoreceptors whose outer segment captures light. The researchers also used a human cell line commonly used in gene therapy studies, as well as an in vivo model carrying mutations in the RHO gene. This gene, which is involved in a form of retinitis pigmentosa for which there is no treatment, encodes a visual pigment present in photoreceptors, rhodopsin. This is the target that has been chosen for this experiment.
To bring the genetic modification to the RHO gene, two delivery strategies were tested. The first is an AAV dual viral vector system containing CRISPR-Cas9, commonly used in gene therapy. The second is an RNP molecular complex containing only the Cas9 protein, which is more difficult to implement but less inflammatory.
With these two strategies, the researchers observed much lower genome editing rates in organoids than those observed in 2D cell lines but consistent with in vivo models. Their results show that in retinal organoids, the outer segments of photoreceptors act as a physical barrier, limiting the entry of genome editing tools into the nucleus. This phenomenon is comparable to that observed in living retinas.
Towards an acceleration of translational research
By showing that organoids "reproduce" the results in vivo, the study validates the use of these "retinal avatars" to serve as a bridge between basic research and the clinic. The results suggest that the success of a gene therapy on organoids would be representative of its success in humans, making it possible to optimize treatments, minimize the use of animal models and accelerate the development of personalized gene therapies. Ultimately, the use of organoids could revolutionize the treatment of inherited retinal diseases, making therapies safer, more effective, and more accessible to patients. It is with this goal in mind that the Institut de la Vision, a pioneer in retinal organoids through several patented protocols, is expanding its culture laboratory.
Take a look at Juliette Pulman's journey at the Institut de la Vision: https://www.linkedin.com/feed/update/urn:li:activity:7427380974075691008
* Juliette Pulman, Hugo Malki, Paul Oudin, Ecem Aydin, Sophie Tran, Laura Visticot, Camille Robert, Anne De Cian, Marie As, Olivier Goureau, Jean-Paul Concordet, Deniz Dalkara, Retinal organoids mirror CRISPR-Cas9 gene editing efficiency observed in vivo, Molecular Therapy Methods & Clinical Development, Volume 33, Issue 4, 2025, 101627, ISSN 2329-0501.
Link to the study: https://www.cell.com/molecular-therapy-family/advances/fulltext/S2329-0501(25)00222-0
